New COVID-19 mRNA Vaccine Candidate Shows Stronger Immune Response and Comparable Safety
New COVID-19 mRNA Vaccine Candidate Shows Stronger Immune Response and Comparable Safety
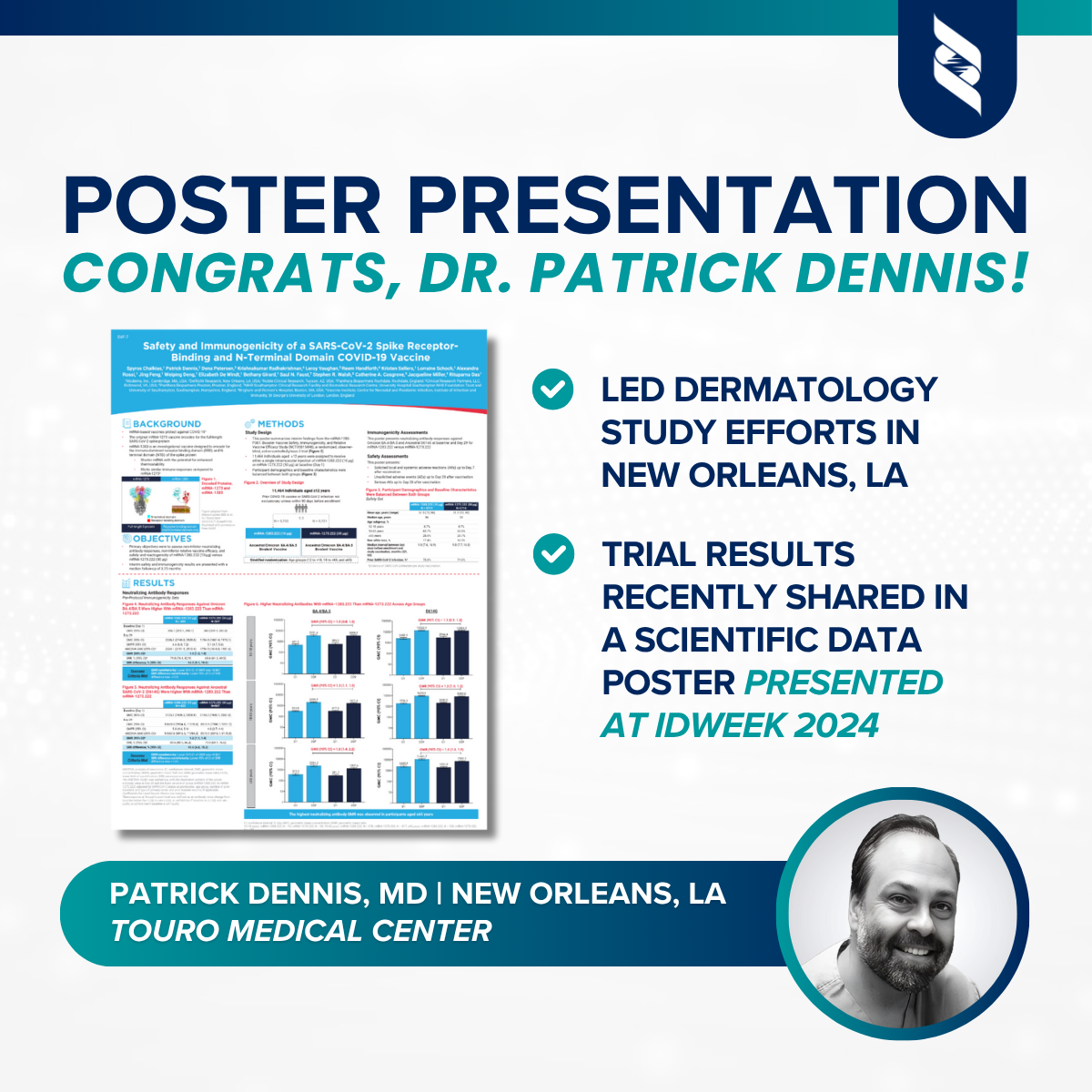
A recent Phase 3 clinical trial evaluating the investigational mRNA-1283.222 vaccine has revealed promising results in the fight against COVID-19. The study compared this new vaccine, developed to target the immunodominant receptor-binding domain (RBD) and N-terminal domain (NTD) of the SARS-CoV-2 spike protein, with the original mRNA-1273.222, which encodes the full-length spike protein.
Unlike its predecessor, mRNA-1283.222 uses a shorter mRNA sequence with the potential for improved stability and manufacturing efficiency. Despite the reduced size, it elicited a stronger immune response, particularly in neutralizing antibody levels against Omicron variants BA.4/BA.5 and the ancestral D614G strain.
Study Design and Population
The randomized, observer-blind, active-controlled trial included 11,464 participants aged 12 years and older. Individuals received a single booster dose of either mRNA-1283.222 (10 µg) or mRNA-1273.222 (50 µg). Demographics and baseline health characteristics were well balanced between the groups, providing a solid foundation for comparison.
Neutralizing antibody levels were measured at baseline and again on Day 29. The goal was to demonstrate that mRNA-1283.222 was at least non-inferior to mRNA-1273.222, criteria the investigational vaccine met and exceeded.
Immunogenicity and Results
Participants who received mRNA-1283.222 developed higher levels of neutralizing antibodies against both Omicron BA.4/BA.5 and ancestral strains compared to those who received the full-length spike protein vaccine. The most significant gains were observed in older adults (65+), an age group at heightened risk for severe COVID-19.
On Day 29, the geometric mean titer (GMC) for Omicron BA.4/BA.5 in the mRNA-1283.222 group was 2,336.2 compared to 1,754.5 in the mRNA-1273.222 group. Similar gains were noted for the D614G variant.
Safety and Tolerability
Importantly, safety outcomes remained similar between the two vaccine arms. Local and systemic side effects, such as fatigue, headache, myalgia, and injection site pain, occurred at comparable rates. Serious adverse events were rare and occurred at nearly identical frequencies in both groups (0.2% vs. 0.3%).
These findings suggest that mRNA-1283.222 offers an enhanced immune response without compromising safety. With its potentially better stability and reduced dosage requirement, this investigational booster may also streamline vaccine distribution and accessibility in the future.
Dr. Patrick Dennis’ Role in Moving Vaccine Science Forward
Dr. Patrick Dennis, principal investigator of DelRicht Research, played an active role in advancing this pivotal research. His involvement in rigorous vaccine trials underscores the importance of site-level leadership in developing next-generation COVID-19 immunizations.
As SARS-CoV-2 continues to evolve, so too must our strategies for protection. Studies like this help inform global public health decisions and ensure that vaccines remain effective across different variants and populations.
To learn more about this trial, view the official study poster presented at the Saudi International Vaccination Forum.
At DelRicht Research, we are happy to congratulate Dr. Patrick Dennis on this publication and are proud to support innovative research that brings meaningful solutions closer to the patients who need them most.
All News